I. Literature Overview
-
Titulus
An aperta iudicium de V, methyltetrahydrofolater in senes depressa aegris - Basic notitia
- Date publication: June MCMXCIII
- Journal: Annales de orci psychiatry
- Research Type: Open-Label Volume Trial
III. Corore Conclusio
De studiis confirmavit quod oris V, methyltetrahydrofolate (mthf) ostendit significant efficaciam in senes tristes aegros, inter XX aegris testimonii DSM-III-r diagnostic criteria, XVI perficitur saltem IV hebdomades de curatio et LXXXI% (XIII aegris) ostendit significant melius in depressive symptoms (Ham-d-XXI score decrevit a ≥50%). Non obvious medicamento actis adversa reactiones Observati in curatione, significans salutem et orci applicationem potential.

II. Detailed Research Design
-
Research Objective
Ad aestimare therapeutica effectus CARDIACUS, repugnans oris mthf (L mg / die) in senes deprimitur aegris et explorandum suum fulsibilitatem ut Tutus interveniente. - Intervention Protocol
- Dosis et Formula: L MG / Day CARDIACUS, repugnans Oral praeparatio (enteric coating technology praesidio activa ingredientia quia recta intestinorum effusio et reducere CARDIACUS acidum damnum).
- Sample characteres: XX senes aegroti (senem 60-82 annis, intelliguntur 68.5 annis), omnes testimonii DSM-III-R Depressio Diagnostic Criteria, Ham-D-XXI score ≥18 et sine comorbid gravibus corporalis Morbi vel mentis perturbationes.
III. Investigatio Tempus
I-septimana placebo washout tempus + VI-septimana aperta, label curatio, cum weekly Assessionem Depressive Symptomata (Ham-D-XXI) et salus Indicatores.
III. Research res gestae et productum valorem
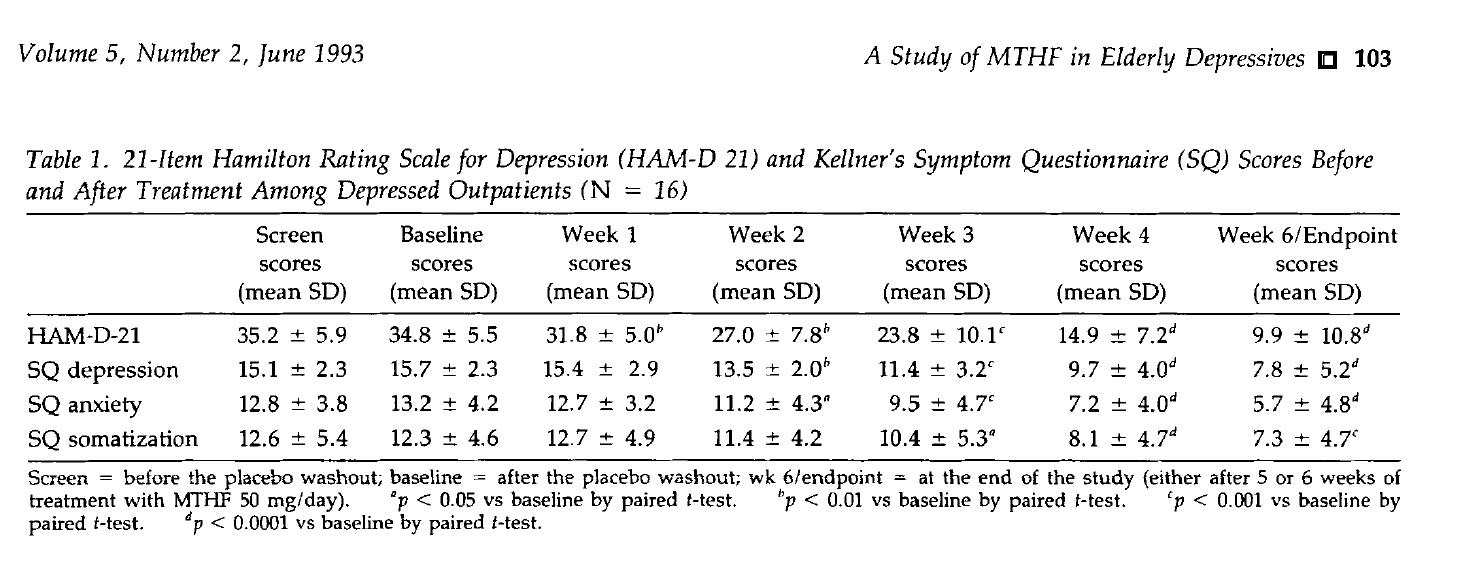
- Efficaciam notitia
- Celeros Celerive: Post VI weeks de curatio, Ham-D-XXI score significantly minuatur a baseline 34.8 ± 5.5 ad 9.9 ± 10.8 (P <0.0001), cum simultaneous remissionem comitante symptoms talem ut anxietatem et somatization.
- Princeps RESPONSIO Rate: LXXXI% of aegris effectum C. RESPONSIO (score diminutio ≥50%), significans quod circa LXXXI de Omnis C senes aegroti potest prodesse.
- Mechanistic Comparatrix: MThf Participat in metabolismi metabolismi, promovet synthesim de s-adenosylmethionine (Eadem), et moderatur metabolismus of neurotransmitters ut dopamine et V-hydroxytryptamine, melior est in mechanism exanimationes incidamus radix (complementary ad target of traditional antidepressants).
2. Salus Testimonium
- Non amet significant abnormitates inventa In Laboratory Indicatores ut sanguinem exercitatione et iecoris / renibus munera Per curatio. Nisi pauci aegris periti mitis non-specifica Symptomata ut capitis et insomnia, sine certis medicamento actis adversa reactiones.
- Product Characteres Magnafonoonim:
- Puritas: ≥99.8%, immunditia Jk12a <0.1%.
- High Safety: "Naturalization Foste", quod productum pervenit ad practice non-toxicus gradu, sine usura toxicus rudis materiae ut formaldehyde et P toluenesulfonic acidum.
- Direct effusio: Non metabolismus requiritur, directe effusio, bioavailability 3-5 temporibus altior quam traditional foste.
3. CHINICAL Significatio
- Aging-Acta advantages: Free a Cardiac Toxicity et orthostatic hypotension metus de traditional antidepressants, Maxime idoneam senes cum reducitur iecoris et renibus munera.
- Combined Application: potest esse ut adiunentiam Therapy pro antidepressants vel sola ad mitis tristitia et Medicamento-intolerant aegris, expanding treatment optiones.
IV. Mechanism Interpretatione et Academic extensio
-
Mechanism de action
Sicut activae Falsum forma in corpore, MTHF afficit depressive pathologiae Per haec meatus:
- Methyl donator munus: participat in centralis nervosi Ratio methylation ponere normalis neurotransmitter synthesis, DNA RECONCINNO, et cellula membrana phospholipid metabolismus.
- Eiusmodi regulantur: promovet cerebrum eiusdem productio, quae agit in methyl donator in methylation modificationem Neurotransmitters ut norepinephrine et V-hydroxytryptamine, Regulating Synaptic Transmissus efficientiam.
- Homocysteine metabolism: Reduces Plasma Homocysteine levels, decrescit eius neurotoxic effectus, et amplio Cognitive-motus ordinandus network.
2. Academic
Support et quod catena
Hoc studium formae est quod clausa loop cum subsequent studiis:
- Synergy combined medication: Passeri et al. (MCMXCI) Duplex-caecus iudicium ostendit quod XV mg / D V, MTHF combined cum Antidepressants auctus ad Ham-D score reductionem a 4.1 puncta comparari Et Monotherapy Group (P <0.01).
- Breakthrough in medicamento, repugnans populatio: A MMXXIII C. Study (NC NCT publice praesto) ostendit quod XV mg / D V, MTHF cum in SSRI melior symptoms in L% de SSRI, repugnans Aegros, praesertim idoneam ad MTHFR Gene C677T Mutationem carriers.
-
Long-term salute verificationem: V, MTHF habet
Bioavailability de LI% -54%, non cumulative toxicity post longum-term usum,
et communis adversa reactiones sunt transeuntes mitis capitis cum incidentiæ
V. Core commoda Magnafonooni Product
|
Dimensio |
Magnafolater® |
Traditional Foste / competitors |
|
Pudicitia |
≥99.8% (HPLC deprehensio, cum Sinis National Health Commission Nuntius No. XIII, MMXVII + USP Signa) |
XCV% -97.9% |
|
Technical obice |
Patented ultrasonic crystallization Technology, stabilis ad locus temperatus pro XLVIII mensibus |
Requirit humilis-temperatus repono, pauper stabilitas |
|
Salus |
Nulla uti toxicus rudis materiae sicut Formaldehyde et P, toluenesulfonic acidum, moderando nocivis impudicitiis talem sicut jk12a et V-methyltetrahydroperoic acidum; Producting productum ad Non-toxicus Level (Shanghai CDC) |
Ut continent genotoxic impurities |
|
Patent layout |
Super XL invention patents covering Crystal formae, praeparatio processus, et applicationem missionibus |
Pauci patents |
VI. Summary of Literature interpretatione
Et pioneering studio in MCMXCIII primo confirmata est V, methyltetrahydrofolater (Magnalfonoonim) habet determinatum efficaciam et salutem in Geriatric tristitia cum mechanism propinqua ad neurotransmitter metabolismus et methylation networks. Et summus puritas rudis materiae et stricte Imperium Imperium, Magnafonoonim Products Exhibeamus maxime princeps salus, dum Optimizing bioavailability et patientes estote obsequio, ita providente humili periculo Et altus adaptive supplementum curatio consilium pro senibus depressa Aegroti, et expectata fiet novum arbitrium in orci curatio.
Market Prospectus: cum global senescens agger Mentis valetudo foro expectat ut pervenire $ 537.97 billion per MMXXV.
Magnafonoeolate cum suis testimonio, secundum commoda est expectata ludere momenti valorem in missionibus ut orci nutritionis Support et eget salutem products.
References: GuardiD GuaaDali Maurizio Fava, Fausto Mazzi et Pietro La Greca. "An aperta iudicium de Methyltetrahydrofolater in senibus depressus patientes estote. " Annales orci psychiatry V, Non. II (MCMXCIII) 101-105. https://Do.org/10.3109/10401239309148970.

 Español
Español Português
Português  русский
русский  Français
Français  日本語
日本語  Deutsch
Deutsch  tiếng Việt
tiếng Việt  Italiano
Italiano  Nederlands
Nederlands  ภาษาไทย
ภาษาไทย  Polski
Polski  한국어
한국어  Svenska
Svenska  magyar
magyar  Malay
Malay  বাংলা ভাষার
বাংলা ভাষার  Dansk
Dansk  Suomi
Suomi  हिन्दी
हिन्दी  Pilipino
Pilipino  Türkçe
Türkçe  Gaeilge
Gaeilge  العربية
العربية  Indonesia
Indonesia  Norsk
Norsk  تمل
تمل  český
český  ελληνικά
ελληνικά  український
український  Javanese
Javanese  فارسی
فارسی  தமிழ்
தமிழ்  తెలుగు
తెలుగు  नेपाली
नेपाली  Burmese
Burmese  български
български  ລາວ
ລາວ  Latine
Latine  Қазақша
Қазақша  Euskal
Euskal  Azərbaycan
Azərbaycan  Slovenský jazyk
Slovenský jazyk  Македонски
Македонски  Lietuvos
Lietuvos  Eesti Keel
Eesti Keel  Română
Română  Slovenski
Slovenski  मराठी
मराठी  Srpski језик
Srpski језик 









 Online Service
Online Service